ISA SPEX Fluorimeter for Quantitating Fluorescent Microsphere Deposition FluoroMax-2
August 1996
General requirements for instrumentation include:
- A reference system to monitor and correct fluctuations in source intensity.
- Variable monochrometers that can select excitation and emission wavelengths with 1-nm increments and the ability to adjust spectral band width to control peak resolution and intensity.
- Linearity over the light spectrum within the expected intensity range. We advise a mean delivery of 1,000 FMS per piece. A minimum delivery of 400 FMS per piece is required for a 95% confidence interval for a blood flow measurement (Buckberg et al., 1991).
- A software package that allows serial intensity measurements on a single sample containing multiple fluorophors with unique excitation, emission and band width requirements. Intensity data should be stored in a form compatible with standard data analysis software.
The linearity of the instruments was measured for a serial dilution of microspheres loaded with pure yellow-green dye. Microsphere concentration ranged from 10 to 2500 microspheres per ml solvent. The linear correlation was excellent, R=0.999 (figure 1).
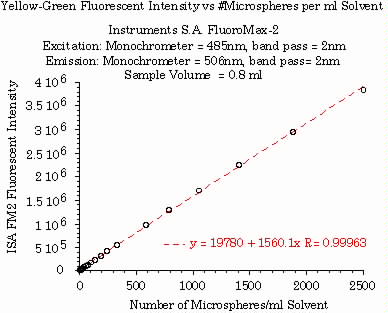
Figure 1.
The performance capabilities of the FluoroMax-2 for measuring ROBF were determined by simultaneous injections of radio nuclides and fluorescent labeled microspheres into an anesthetized pig including: Scandium46 (4.3x106), Tin113 (4x105), Orange (2x106) and Yellow-green(2x105) microspheres. The number of Tin and Yellow-Green microspheres were an order of magnitude lower than normal, to test the sensitivity of the fluorimeter at low signal intensities. One hundred pieces of lung tissue were selected to validate measured fluorescence against radio nuclides. Radioactive intensities were determined and the lung tissue soaked in 2-ethoxyethyl acetate for 48 hours. Fluorescent intensities were measured using the CWA program.
Correlation between Orange fluorescent intensity and Scandium46 was excellent, R=0.986 (figure 2). Mean fluorescent intensity for Orange (4.935x107) is equivalent to 400 FMS per piece, the minimum necessary for 95% confidence interval.
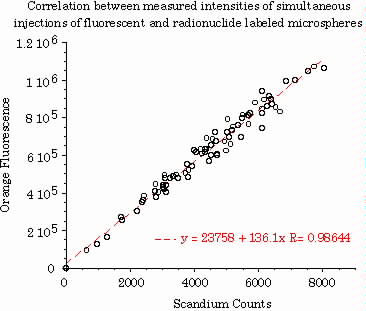
Figure 2.
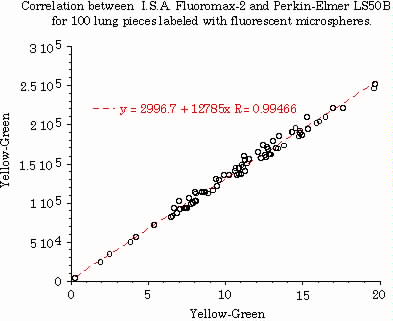
Figure 3.
The software control of the FluoroMax-2 is well thought-out in terms of ease and versatility of operation. Experimental parameters are easy to set up and store for future acquisitions. The instrument status page is particularly convenient in terms of monitoring and modification of the instrument parameters, without having to exit a data acquisition mode.
References:
- Buckberg, G.D., J.C. Luck, D.B. Payne, J.I.E. Hoffman, J.P. Archie and D.E. Fixler. Some sources of error in measuring regional blood flow with radioactive microspheres. J Appl Physiol. 74:2585-97 1993.
- Glenny, R.W., S. Bernard and M.Brinkley. Validation of Fluorescent-labeled microsphere for measurement of regional organ perfusion. J Appl Physiol. 74:2585-97 1993.